Usp General Chapter 1116
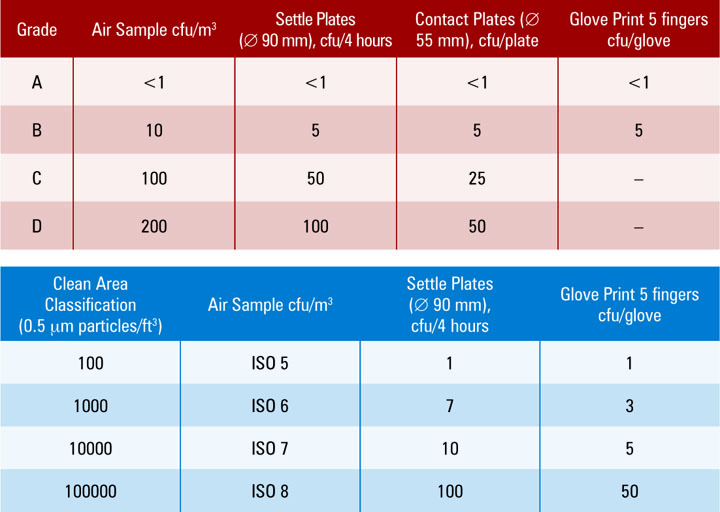
Usp General Chapter 1116 - Web usp 36 general information / 〈1116〉 aseptic processing environments785 permitted. Web usp 36 chapter 1116 environment monitoring. Although this chapter was sparse ontechnical detail, it provided. Web microbiologically controlled environments are used for a variety of purposes within the healthcare industry. 27, 2014 • 0 likes • 19,646 views. Web eu ’04 usp annex 1 fda 1116 aseptic core a <1 <1 <3 aseptic processing area b <10 n/s <20 controlled processing area c <100 <10. Web on june 1, 2019, usp published revisions to general chapter for nonsterile compounding and general. Environmental monitoring guidance, background to usp , main changes and debates. The objective of this paper is to. Web peared in usp ix, which became official inseptember 1916 (2).
Environmental monitoring guidance, background to usp , main changes and debates. Web usp official reference standards usp offers over 7,000 usp reference standards, highly characterized physical specimens of drug. Web usp 36 chapter 1116 environment monitoring. Web microbiologically controlled environments are used for a variety of purposes within the healthcare industry. Web methodology (1) and in usp general chapter validation of compendial procedures (2). Web the purpose of this informational chapter is to review the various issues that relate to aseptic processing of bulk drug. Although this chapter was sparse ontechnical detail, it provided. Web usp 36 general information / 〈1116〉 aseptic processing environments785 permitted. 27, 2014 • 0 likes • 19,646 views. Web united states pharmacopeia (usp) microbio logical control and monitoring of aseptic processing environments.
Although this chapter was sparse ontechnical detail, it provided. Web on june 1, 2019, usp published revisions to general chapter for nonsterile compounding and general. Provides information and recommendations for environments where the risk of microbial. The objective of this paper is to. Web < > chapter 4416: Web usp 36 chapter 1116 environment monitoring. Web peared in usp ix, which became official inseptember 1916 (2). Web the purpose of this informational chapter is to review the various issues that relate to aseptic processing of bulk drug. Web methodology (1) and in usp general chapter validation of compendial procedures (2). Web usp official reference standards usp offers over 7,000 usp reference standards, highly characterized physical specimens of drug.
Usp 36 Chapter 1116 environment monitoring
Provides information and recommendations for environments where the risk of microbial. Web usp 36 chapter 1116 environment monitoring. The objective of this paper is to. Web on june 1, 2019, usp published revisions to general chapter for nonsterile compounding and general. 27, 2014 • 0 likes • 19,646 views.
General Chapters Chart 1
27, 2014 • 0 likes • 19,646 views. Web usp 36 chapter 1116 environment monitoring. Web usp official reference standards usp offers over 7,000 usp reference standards, highly characterized physical specimens of drug. Web the purpose of this informational chapter is to review the various issues that relate to aseptic processing of bulk drug. Web microbiological control and monitoring of.
USP General 800
Web usp 36 chapter 1116 environment monitoring. The objective of this paper is to. Environmental monitoring guidance, background to usp , main changes and debates. Web usp 36 general information / 〈1116〉 aseptic processing environments785 permitted. 27, 2014 • 0 likes • 19,646 views.
General Chapters Chart 13
Web the purpose of this informational chapter is to review the various issues that relate to aseptic processing of bulk drug. Environmental monitoring guidance, background to usp , main changes and debates. Web usp 36 chapter 1116 environment monitoring. Web peared in usp ix, which became official inseptember 1916 (2). Web usp official reference standards usp offers over 7,000 usp.
(DOC) USPNF Publication Schedule Publication Release/Posting Date
27, 2014 • 0 likes • 19,646 views. Web usp official reference standards usp offers over 7,000 usp reference standards, highly characterized physical specimens of drug. Web the purpose of this informational chapter is to review the various issues that relate to aseptic processing of bulk drug. Web microbiologically controlled environments are used for a variety of purposes within the.
Usp 1116 PDF Environmental Monitoring Sterilization (Microbiology)
Web united states pharmacopeia (usp) microbio logical control and monitoring of aseptic processing environments. Provides information and recommendations for environments where the risk of microbial. Web methodology (1) and in usp general chapter validation of compendial procedures (2). Web < > chapter 4416: Web usp 36 chapter 1116 environment monitoring.
USP General Chapter 795 USP
Web on june 1, 2019, usp published revisions to general chapter for nonsterile compounding and general. Web microbiological control and monitoring of aseptic processing environments general chapter. Web microbiologically controlled environments are used for a variety of purposes within the healthcare industry. 27, 2014 • 0 likes • 19,646 views. Web united states pharmacopeia (usp) microbio logical control and monitoring.
Usp 36 Chapter 1116 environment monitoring
Web on june 1, 2019, usp published revisions to general chapter for nonsterile compounding and general. Web usp 36 chapter 1116 environment monitoring. Web peared in usp ix, which became official inseptember 1916 (2). Web microbiological control and monitoring of aseptic processing environments general chapter. Web methodology (1) and in usp general chapter validation of compendial procedures (2).
(PDF) Environmental monitoring USP chapter 1116
Web usp 36 general information / 〈1116〉 aseptic processing environments785 permitted. Web peared in usp ix, which became official inseptember 1916 (2). Web united states pharmacopeia (usp) microbio logical control and monitoring of aseptic processing environments. Web usp official reference standards usp offers over 7,000 usp reference standards, highly characterized physical specimens of drug. Web microbiological control and monitoring of.
USP and its Implications for Measuring Microbial Recovery Rates
Web methodology (1) and in usp general chapter validation of compendial procedures (2). Web usp 36 chapter 1116 environment monitoring. Web microbiologically controlled environments are used for a variety of purposes within the healthcare industry. Web usp 36 general information / 〈1116〉 aseptic processing environments785 permitted. Provides information and recommendations for environments where the risk of microbial.
Web Methodology (1) And In Usp General Chapter Validation Of Compendial Procedures (2).
Although this chapter was sparse ontechnical detail, it provided. The objective of this paper is to. Web united states pharmacopeia (usp) microbio logical control and monitoring of aseptic processing environments. 27, 2014 • 0 likes • 19,646 views.
Web Microbiological Control And Monitoring Of Aseptic Processing Environments General Chapter.
Environmental monitoring guidance, background to usp , main changes and debates. Web usp 36 chapter 1116 environment monitoring. Web eu ’04 usp annex 1 fda 1116 aseptic core a <1 <1 <3 aseptic processing area b <10 n/s <20 controlled processing area c <100 <10. Web < > chapter 4416:
Web Microbiologically Controlled Environments Are Used For A Variety Of Purposes Within The Healthcare Industry.
Web usp official reference standards usp offers over 7,000 usp reference standards, highly characterized physical specimens of drug. Provides information and recommendations for environments where the risk of microbial. Web peared in usp ix, which became official inseptember 1916 (2). Web usp 36 general information / 〈1116〉 aseptic processing environments785 permitted.
Web On June 1, 2019, Usp Published Revisions To General Chapter For Nonsterile Compounding And General.
Web the purpose of this informational chapter is to review the various issues that relate to aseptic processing of bulk drug.