What Kinds Of Elements Form Covalent Bonds
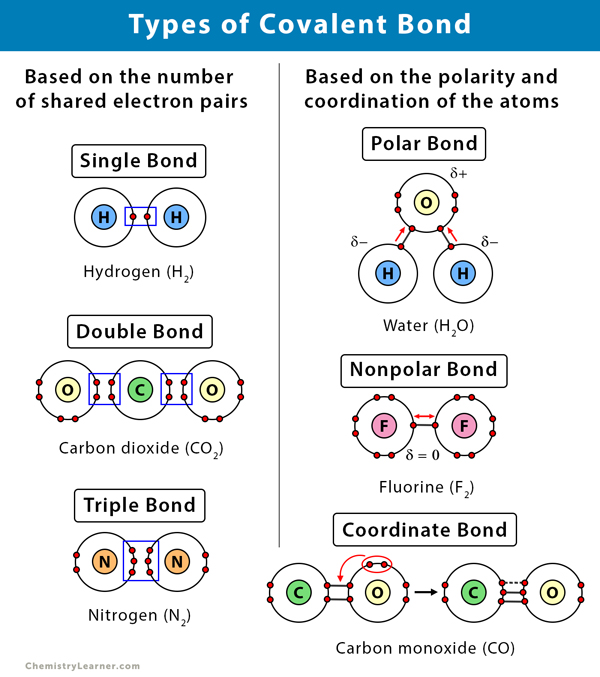
What Kinds Of Elements Form Covalent Bonds - There are two types of covalent bonds: Both the atomic nuclei of the shared atom together attract these. Web which elements form covalent bonds? For example, the hydrogen molecule, h 2, contains a covalent bond. Web details of 2.1: Web google classroom chemical bonds hold molecules together and create temporary connections that are essential to life. Atoms have two ways to achieve an octet. Web covalent bonds take place between nonmetal elements in the periodic table such as carbon, hydrogen, oxygen, and nitrogen. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms. Web covalent bond involves the sharing of electrons between atoms.
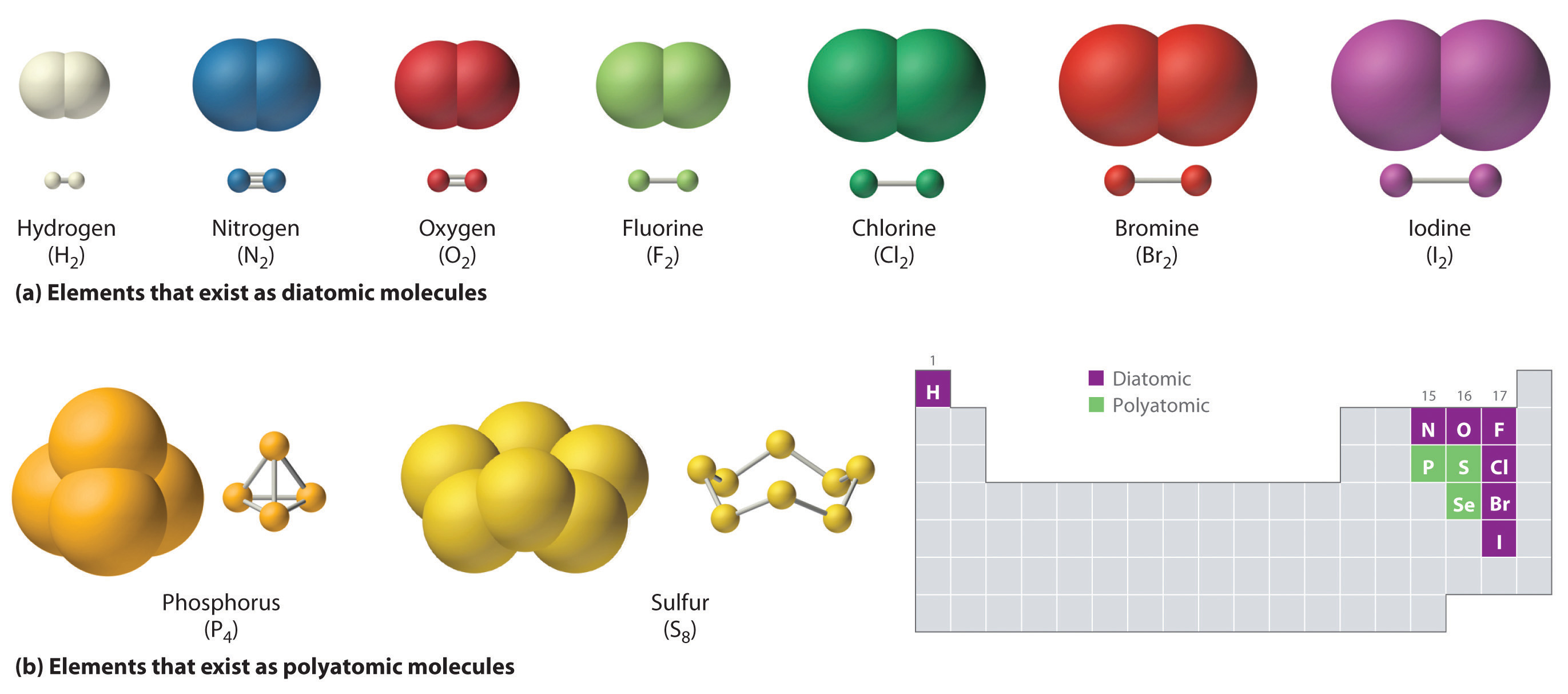
Both the atomic nuclei of the shared atom together attract these. Therefore, atoms which make covalent bonds can not be either give electrons or accept. Web covalent bonds take place between nonmetal elements in the periodic table such as carbon, hydrogen, oxygen, and nitrogen. Covalent bonds form between atoms of nonmetallic elements. Nonpolar covalent bonds form between two. Web google classroom chemical bonds hold molecules together and create temporary connections that are essential to life. Web starting with fluorine as the most electronegative, electronegativity decreases as it moves down and left on the periodic table, reaching cesium, the least. The pair of shared electrons. Atoms have two ways to achieve an octet. Types of chemical bonds including covalent, ionic,.
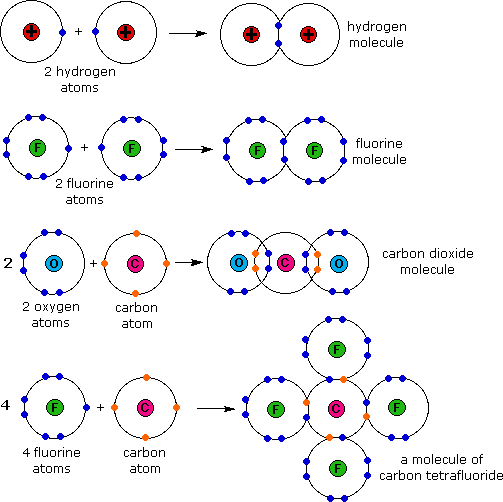
For example, the hydrogen molecule, h 2, contains a covalent bond. Web covalent bonds form when electrons are shared between two nonmetals. Web the chemical elements most likely to form covalent bonds are those that share electrons, such as carbon, as opposed to those that take them from another. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms. Web details of 2.1: Nonmetal atoms frequently form covalent bonds with other nonmetal atoms. Bonds within most organic compounds are described as. Atoms have two ways to achieve an octet. Web which elements form covalent bonds? Web what types of elements will you not find in a covalent bond?
Covalent Bond Definition, Types, and Examples
There are two types of covalent bonds: Web read discuss a covalent bond is formed when two atoms exchange one or more pairs of electrons. Web covalent bonds form when electrons are shared between two nonmetals. Web details of 2.1: Types of chemical bonds including covalent, ionic,.
Types of chemical bonding diagram. Covalent polar and nonpolar, ionic
Web the octet rule causes atoms to form bonds in such a way that they acquire eightvalence electrons. Atoms have two ways to achieve an octet. Metals, metalloids, and noble gases. Web what types of elements will you not find in a covalent bond? Web apr 1, 2014.
10 Notable Differences Between Ionic And Covalent Bonds Current
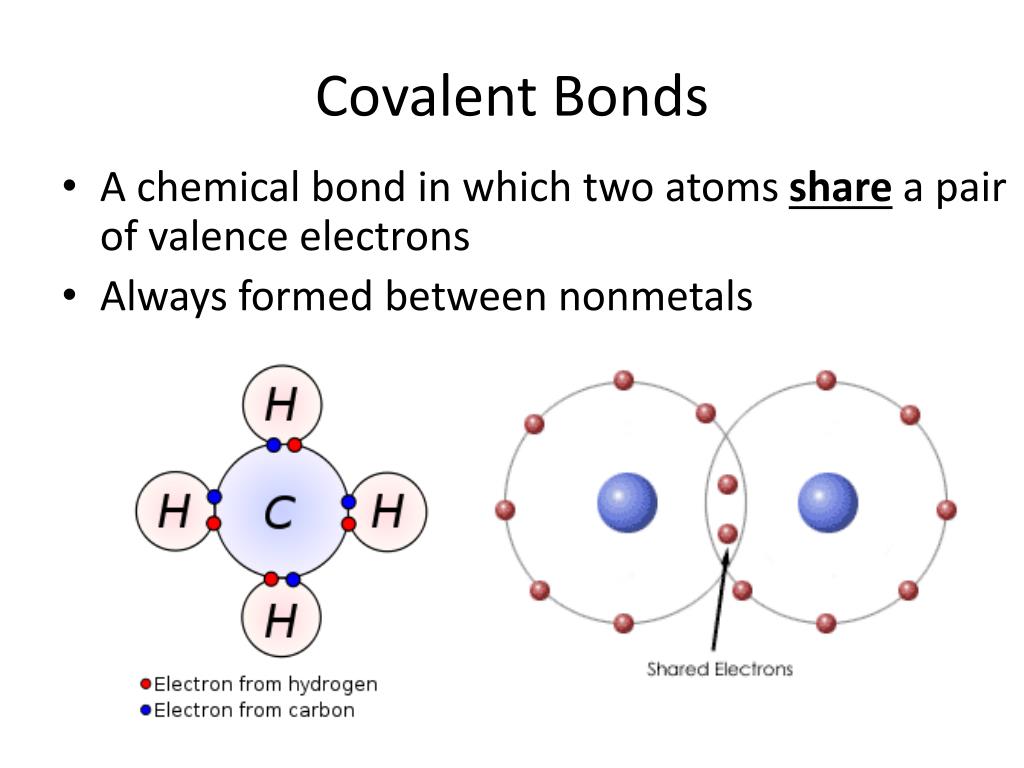
Web google classroom chemical bonds hold molecules together and create temporary connections that are essential to life. Web forming a covalent bond a covalent bond is formed when two atoms share a pair of electrons. Web formation of covalent bonds. Web covalent bond involves the sharing of electrons between atoms. What types of elements form covalent compounds and.
PPT Covalent Bonds PowerPoint Presentation, free download ID6647183
Web formation of covalent bonds. Web forming a covalent bond a covalent bond is formed when two atoms share a pair of electrons. Types of chemical bonds including covalent, ionic,. There are two types of covalent bonds: What types of elements form covalent compounds and.
EduMission Chemistry Form 4 Chapter 5 Covalent Bond
Metals, metalloids, and noble gases. Bonds within most organic compounds are described as. Web details of 2.1: There are two types of covalent bonds: Therefore, atoms which make covalent bonds can not be either give electrons or accept.
2.2A Covalent Bonds and Other Bonds and Interactions Medicine LibreTexts
Types of chemical bonds including covalent, ionic,. Web starting with fluorine as the most electronegative, electronegativity decreases as it moves down and left on the periodic table, reaching cesium, the least. Web forming a covalent bond a covalent bond is formed when two atoms share a pair of electrons. Web covalent bonds form when electrons are shared between two nonmetals..
Explain what a covalent bond is, what types of elements form covalent
Web covalent bond involves the sharing of electrons between atoms. Therefore, atoms which make covalent bonds can not be either give electrons or accept. Web starting with fluorine as the most electronegative, electronegativity decreases as it moves down and left on the periodic table, reaching cesium, the least. Both the atomic nuclei of the shared atom together attract these. Nonmetal.
Which elements form a covalent bond? Quora
Web the octet rule causes atoms to form bonds in such a way that they acquire eightvalence electrons. What types of elements form covalent compounds and. For example, the hydrogen molecule, h 2, contains a covalent bond. Metals, metalloids, and noble gases. When two nonmetal atoms bond, they mutually.
CH150 Chapter 4 Covalent Bonds and Molecular Compounds Chemistry
Web forming a covalent bond a covalent bond is formed when two atoms share a pair of electrons. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms. Covalent bonds form between atoms of nonmetallic elements. Nonpolar covalent bonds form between two. Web the octet rule causes atoms to form bonds in.
Covalent Bonding (Biology) — Definition & Role Expii
What types of elements form covalent compounds and. Web read discuss a covalent bond is formed when two atoms exchange one or more pairs of electrons. When two nonmetal atoms bond, they mutually. Web starting with fluorine as the most electronegative, electronegativity decreases as it moves down and left on the periodic table, reaching cesium, the least. Web which elements.
Web The Octet Rule Causes Atoms To Form Bonds In Such A Way That They Acquire Eightvalence Electrons.
Web which elements form covalent bonds? Web covalent bonds take place between nonmetal elements in the periodic table such as carbon, hydrogen, oxygen, and nitrogen. For example, the hydrogen molecule, h 2, contains a covalent bond. A covalent bond involves a pair of electrons being shared between atoms.
Web Forming A Covalent Bond A Covalent Bond Is Formed When Two Atoms Share A Pair Of Electrons.
Web covalent bond involves the sharing of electrons between atoms. Web details of 2.1: Covalent bonds form between atoms of nonmetallic elements. In general, bonds are considered to be covalent if the electronegativity difference.
What Types Of Elements Form Covalent Compounds And.
When two nonmetal atoms bond, they mutually. The pair of shared electrons. There are two types of covalent bonds: Metals, metalloids, and noble gases.
Nonpolar Covalent Bonds Form Between Two.
Types of chemical bonds including covalent, ionic,. Both the atomic nuclei of the shared atom together attract these. Bonds within most organic compounds are described as. Web starting with fluorine as the most electronegative, electronegativity decreases as it moves down and left on the periodic table, reaching cesium, the least.